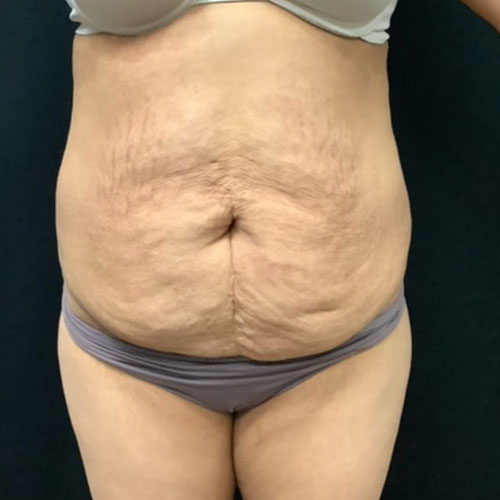
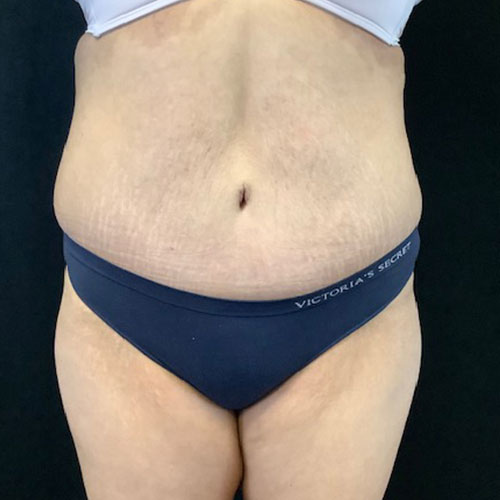
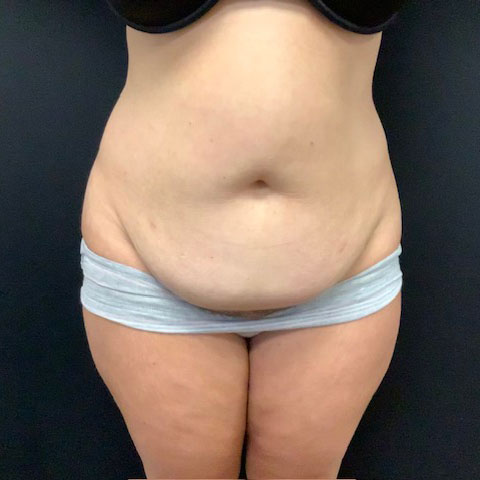
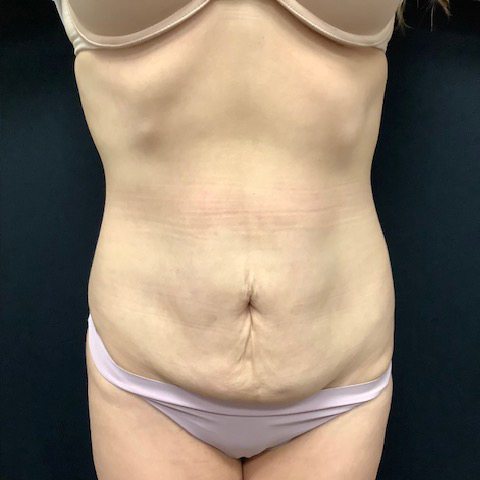
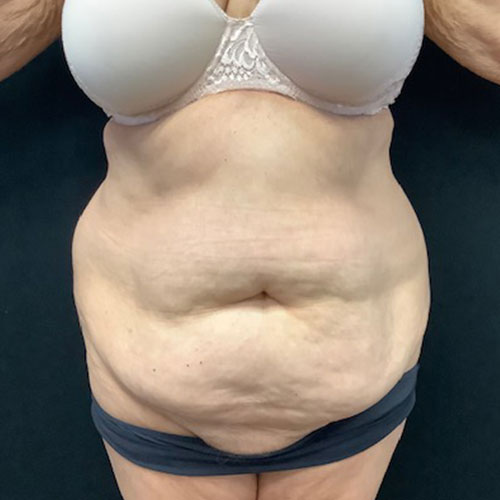
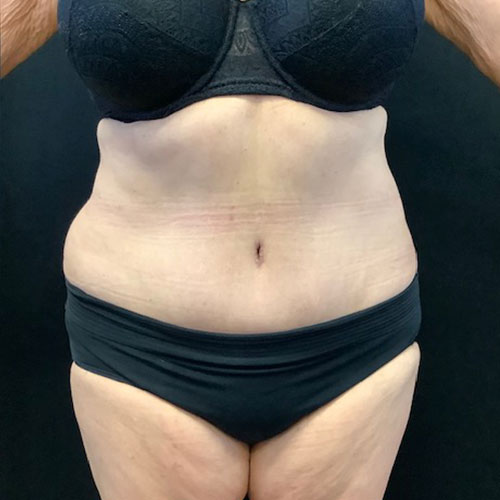
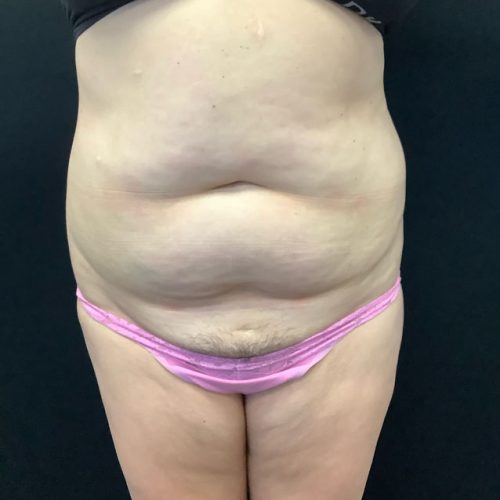
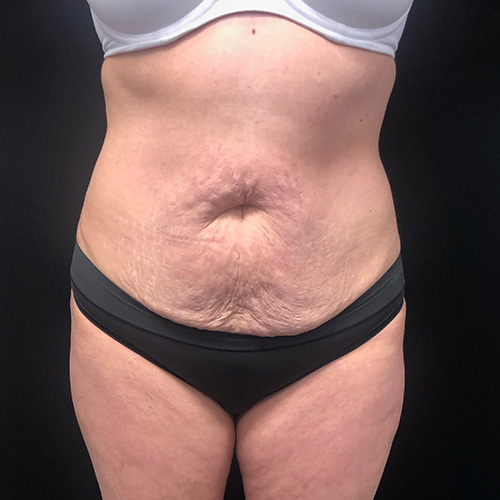
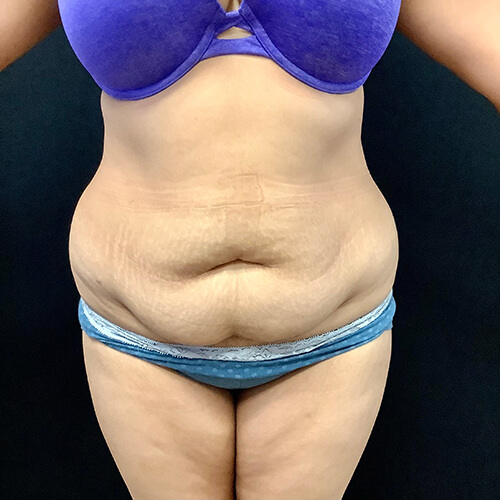
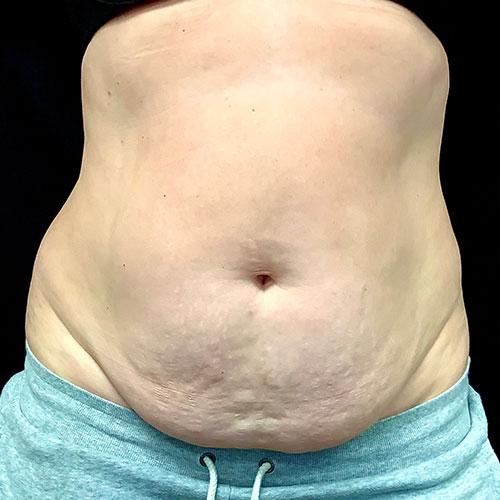
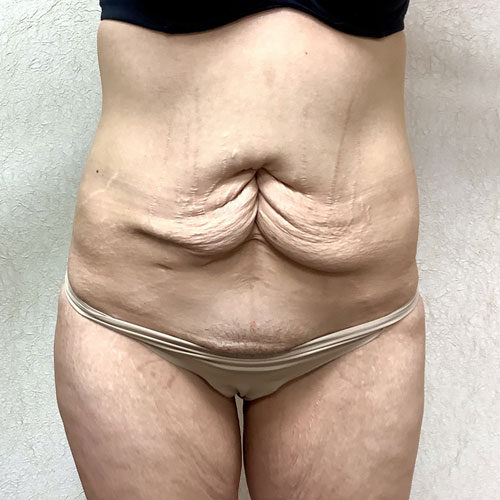
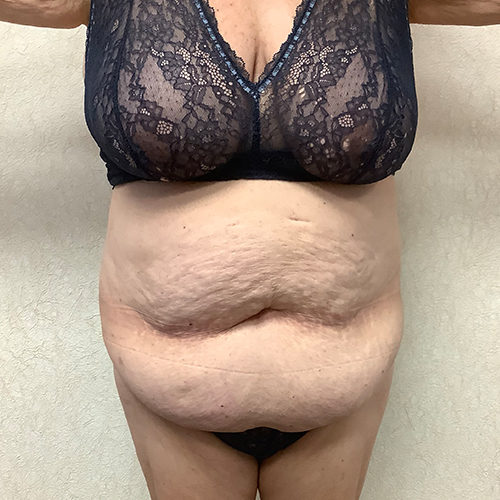
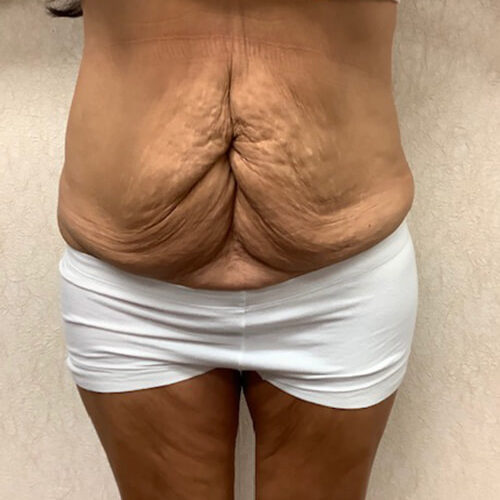
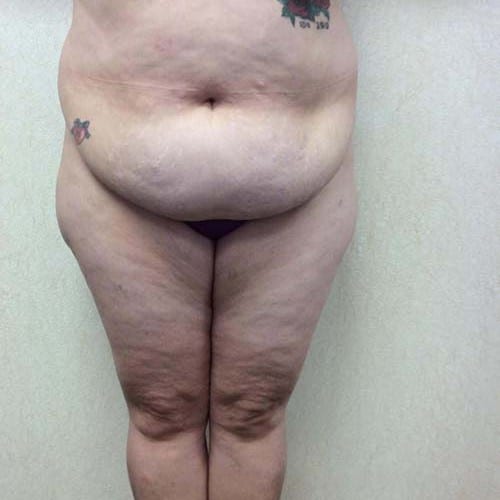
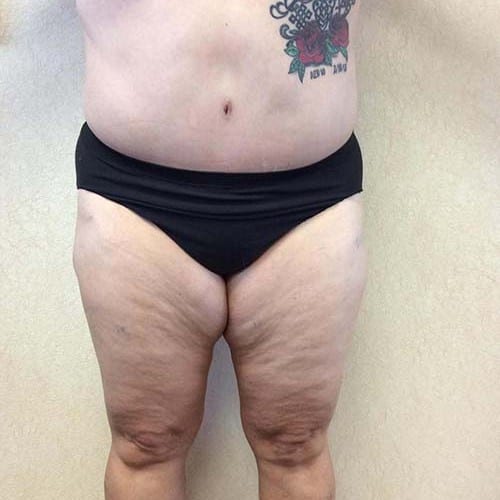
Dr. Wald Looks forward to
helping you reach your
cosmetic goals.
Schedule a consultation
The aesthetic institute
Robert Wald, MD
100 E. Valencia Mesa Drive, Suite 300
Fullerton, CA 92835
Phone: 714-312-3549
Fax: 714-738-1862